Business opportunity
About 50% of human epidermal growth factor receptor 2 (HER2)-positive tumors express p95HER2, a known constitutively hyperactive truncated version of HER2. This HER2-variant gives aggressive disease, inferior prognosis, and resistance to current HER2-directed therapies. There is currently no diagnostic assay available to identify the patients with p95HER2 expression, and no therapy targeting p95HER2. The scientists have addressed this medical need by developing a novel diagnostic antibody and Chimeric Antigen Receptor (CAR) T cell therapy for p95HER2 positive breast cancer. The diagnostic antibody may be applicable to all HER2+ breast cancer patients. This CART may offer a treatment for patients suffering from p95HER2 positive metastatic breast cancer, a group of patients with poor prognosis and high unmet need.
Inven2 is seeking to out-license the patent application to pharmaceutical companies and/or biotechnology SMEs or partners for co-development of the technology. The scientific group is also developing STEAP1 CARs for metastatic prostate cancer.
Technology description
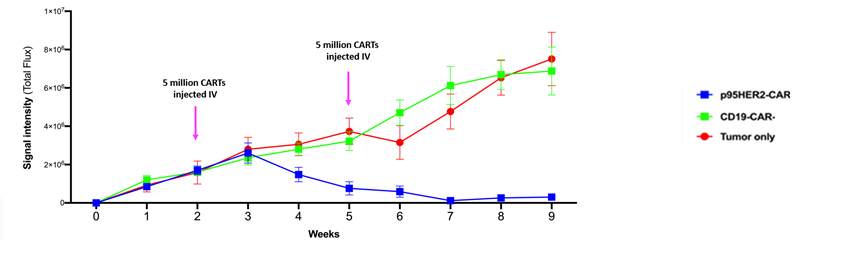
The novel antibody has a high affinity and specificity and can be used in different techniques for diagnostic purposes. The antigen-binding unit is demonstrated suitable for implementation into CARs, and retains sufficient affinity and specificity for p95HER2 in this format. The CARs are well expressed in T cells, and have potent anti-tumour functionality in vitro. Further, their therapeutic effect is demonstrated by in vivo experiments.
Advantages
The data demonstrates an effective and highly significant reduction in tumour mass once the CARTs are injected by systemic i.v. administration. They have shown this in a preclinical mouse model of human breast cancer.
IPR
A patent application has been filed.

