BUSINESS OPPORTUNITY
Lipopolysaccharides (LPS) or endotoxins are an important part of the outer membrane of Gram-negative bacteria. When endotoxins enter the body, they can cause very strong immune reactions that can lead to endotoxemia, sepsis, and toxic shock. It is therefore pertinent that pharmaceutical products and medical equipment that enter the body are endotoxin free. Sensitive detection and efficient removal are therefore important and of high value.
The current gold standard for LPS detection is the Limulus amoebocyte lysate (LAL) assay.Researchers connected to the University of Oslo have identified a peptide that binds LPS at picomolar affinity. This peptide can be used to detect LPS with a sensitivity comparable or better than the LAL assay, and it can detect all LPS species tested. Current removal methods include for example the use of Polymyxin B, which has a much lower affinity for LPS compared to our peptide. The peptide could thus potentially also strongly improve the removal efficacy, e.g. when coupled to solid supports.
We are interested in industrial partners for licensing and further development of the technology.
TECHNOLOGY DESCRIPTION
Detailed analysis of the interaction of LPS with the peptide has revealed that:
- The peptide binds to the conserved lipid A part of LPS.
- It binds broadly to LPS from different species, including B. henselae, N. lactamica, P. gingivalis, S. typhyrium, V. cholerae, and E. coli.
- The affinity (dissociation constant) is approximately 60 – 70 pM. This is several orders of magnitude lower (stronger binding) than any known LPS binder (Toll-like receptor 4 – 141 µM, CD14 – 74nM, MD-2 – 2.33 µM, LBP – 3.5 nM, polymyxin B – 46 µM).
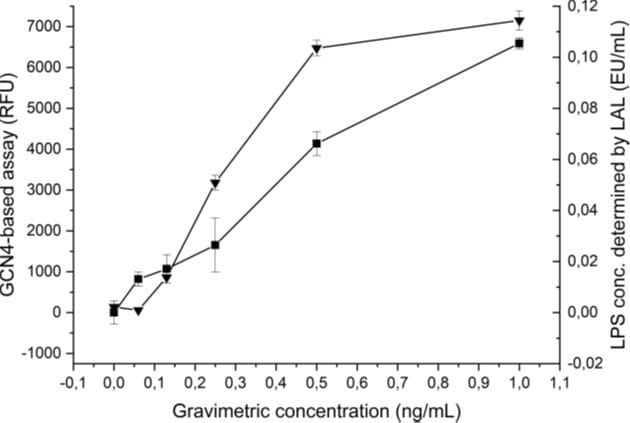
- Benchmarking to the LAL assay demonstrates that the detection threshold is at least similar than the LAL-assay (~0.062 ng/ml): triangles: LAL-based, squares: peptide-based.
- The peptide also “dissolves” LPS-micelles, which may have relevance for known LPS masking effects.
PUBLICATIONS
A selection of data has been published in Hatlem et al., (2023) “A trimeric coiled-coil motif binds bacterial lipopolysaccharides with picomolar affinity”.
INTELLECTUAL PROPERTY
A patent application (WO2021260144A1) is in national phase.
